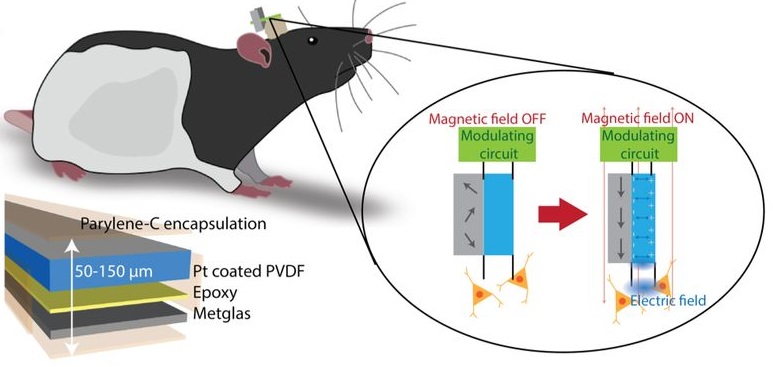
Our solution...
We have successfully created magnetoelectric devices
capable of transforming external magnetic fields to
controllable electric fields strong enough to wirelessly
stimulate targeted neural regions in freely moving rats
without genetic modification. By coupling a
piezoelectric and magnetostrictive material at an
acoustic resonance, the application of an external
magnetic field allows for the stimulation of cells,
both in vivo and in vitro. Having been able to
successfully power implanted electrodes in freely moving
rats, we demonstrate the ability to wirelessly modulate
neural, and thus behavioral, activity. In contrast to
traditional inductive coupling, magnetoelectric
materials are scalable and remain capable of generating
large voltages with a small device footprint. Our
results provide the groundwork to create much smaller
implantable devices for neural modulation in the near
future.
My involvement in this project, led by Amanda Wickens of
the Robinson Lab, has been the design and manufacture of
an implant for both in vivo and in vitro testing, as
well as the analysis of rodent behavior using a version
of DeepLabCut, which can be found on my GitHub
here.